Cool Jobs: Like Mother Nature
Scientists observe what works best for animals, plants in efforts to build better products
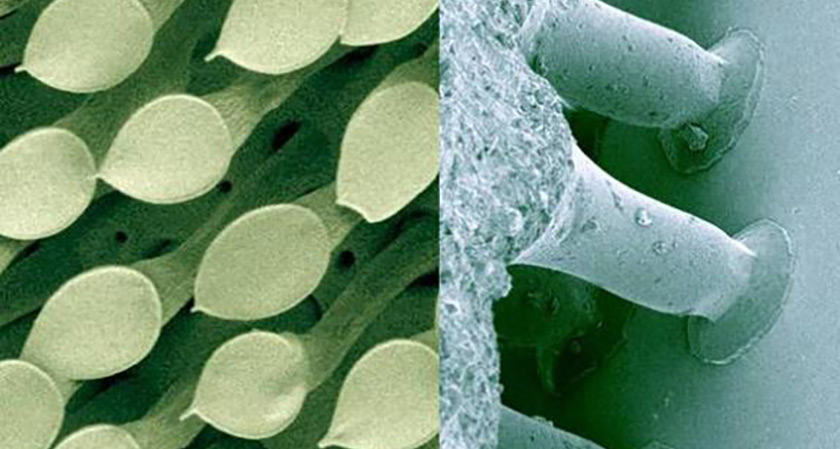
Revealed under a microscope, the hairs on a beetle’s foot (left) were the inspiration for a supersticky tape created by Stanislav Gorb (right).
Stanislav Gorb and Gottlieb Binder GmbH
Imagine a tape so strong it can hold your weight and be peeled off thousands of times without losing its stickiness.
Stanislav Gorb, a professor of zoology at the University of Kiel in Germany, figured out how to make such a tape last year. His creation was made possible by biomimicry, the science of looking to nature for inspiration in designing new technology.
Over the course of billions of years, living things have evolved through a process of trial and error. Many scientists now believe that research in the lab can benefit from studying what does and doesn’t work in nature.
Sticky feet, sticky tape
For his supersticky tape, Gorb borrowed an idea from flies, spiders, beetles and geckos. Their feet have hundreds of thousands of tiny hairs that grab surfaces, allowing them to walk on walls and ceilings.
To see which of these creatures excelled at clinging to surfaces, Gorb put them on a flat disk and spun it. It turns out that male leaf beetles are particularly good at staying put.
So Gorb took a close look at the hairs on these beetles’ feet and found they had flat tips that spread out, allowing the insects to better grab surfaces. The hairs’ sticking power had nothing to do with glue. Instead, the beetles cling to surfaces just like a raindrop clings to a window. What allows both to keep from falling off is the attraction between molecules, something called the Van der Waals force. This force is weak, though, so when a raindrop gets heavy enough, it loses its grip and slides down.
Because beetle feet have hundreds of thousands of hairs — each hair exerting the Van der Waals force — they can stick to surfaces for as long as they want.
For their prototype, Gorb and his team created a tape patterned with similar tiny hairs made from silicone. Silicone, a compound that stands up well to heat and water, is often used to make tape. When the researchers put their new tape to the test, they discovered that a piece slightly less than 20 centimeters by 20 centimeters (or 8 inches on each side) can hold the weight of an adult man.
Today, the new tape, produced in collaboration with the German company Gottlieb Binder, is the first glue-free, reversible type on the market. (Velcro doesn’t count because it has hooks on one side, loops on another. Also, Velcro won’t hold your weight.)
After years of studying what Gorb describes as “animal structures and their remarkable abilities,” he says it was gratifying to be able to apply the discovery about beetle feet. But he couldn’t have done it without bringing together a good understanding of many different subjects.

His Ph.D. in biology and entomology (the study of insects) gave him a huge head start. But it wasn’t enough, he says. He needed “biology and a little bit of everything — physics, chemistry, engineering, design.” When he couldn’t do all the research himself, he hired other scientists with the necessary expertise to pitch in.
Flipper science
Collaboration is familiar ground for zoologist Frank Fish, who also worked with a variety of scientists to come up with his remarkable biomimicry application.
The Pennsylvania scientist at West Chester University discovered something important about how water flows over a humpback whale’s flippers. But it took Fish years to figure out exactly what was going on and how to harness the flippers’ design to make and conserve energy.
The story begins one day in the early 1980s, when Fish was browsing through a Boston art store. Fish is an expert in animal biomechanics, the study of how bodies move. In the store, he saw a sculpture of a humpback whale that made him laugh out loud. “I was cocky,” he recalls. “I’d been studying hydrodynamics, and I looked at it and said, ‘The artist got it wrong.’” (Hydrodynamics is the study of liquids in motion.)
The sculpture showed bumps, known as tubercles, along the leading edge of the whale’s flippers. Fish figured that couldn’t be right — those bumps would disrupt the water flow around the flippers, making it hard for the whale to swim.
But when the store owner showed him a photo of a humpback whale, sure enough, there were bumps along the flippers’ leading edges.
“I felt embarrassed and dumb,” says Fish. And then he began wondering what purpose those bumps might serve.
So he called a friend at the Smithsonian Institution in Washington, D.C., to ask if he could have a flipper the next time museum scientists found a dead humpback. Years went by. Eventually, the Smithsonian researchers found a beached humpback.
Fish drove to the New Jersey shore to pick up the animal’s flipper. However, the whale was bigger — much bigger — than he expected. So the scientist had to saw the whale’s 10-foot-long, 300-pound flipper into three sections before hauling each piece into the trunk of his car, which sagged under the weight.
“I had to drive back through New Jersey hoping a policeman wasn’t going to stop me for a safety violation and find rotting body parts in the back of my car,” Fish laughs.
Fish then put the flipper chunks in a freezer. And they stayed there for years while he searched in vain for someone who could make a model of them. Finally, he found a particularly gifted student who helped him analyze the flipper in cross-sections to determine its shape. (The cross-section of an object is a slice that goes all of the way through. When you cut a cucumber into little round pieces, each of those pieces is a cross-section of the vegetable.)
After they digitized that information — converted the data into digital information that a computer could use — an engineer used special software to design a 3-D computer model of the flipper. With the help of a special cutting machine, Fish then used that computer model to cut a real model out of a plastic block. Only then could he analyze it, again with help from another expert.
Flipper lift
Fish next enlisted the help of engineers at the U.S. Naval Academy. When they put the flipper models into a wind tunnel, they discovered the tubercles guided the airflow so that it moved along the flippers’ surface.

To understand why this is important, Fish suggests you put your hand out a car window while travelling. Start with your palm facing the ground, and then rotate your hand by lifting your thumb up.
You will feel two forces — a backward push and an upward lift. If you keep increasing the angle of your hand, the lift force will increase. The lift force moves your hand upward. But only to a point, says Fish.
“What happens as your hand is [tilted] at too high an angle — what’s called the angle of attack — is you stall. And then all you feel is a lot of drag and your hand stops going up,” he says. In fact, it falls down.
That’s because the air is no longer guided along the back of your hand. When your hand is at too high of an angle, the air can`t get around the leading edge of your hand — the thumb side — and adhere to the back of your hand.
“As a result, the air shoots off prematurely and disrupts the pressure differences between the upper and lower surfaces of your hand. And you lose lift,” Fish explains.
In the ocean, tubercles force water to flow so that it is guided along the surface of the whale’s flippers. This means the whale can position its flippers at a sharper angle and get more lift — and therefore more energy — as the water flows over them.
Of course, the design has the same effect on airflow. So Fish set up a Toronto-based company called WhalePower, which is now testing wind turbine blades designed with those tubercle-like bumps.

WhalePower has also licensed this design to another company that is using it to build industrial fans for warehouses and barns. The fans use half the number of blades, move 25 percent more air and use 25 percent less power than fans with conventional blades turning at the same speed.
Fish says none of this would have been possible without the help of engineers designing experiments to figure out how the flippers worked and mathematicians creating computer models to test what size and number of tubercles worked best.
The solar leaf
Harvard University chemist Daniel Nocera also credits the work of many collaborating scientists for his advanced solar cell. A solar cell is a device that converts the energy from sunlight into electrical energy. Nocera refers to his creation as an artificial leaf. Plants harness energy through a process called photosynthesis in which sunlight is used to drive a host of chemical reactions. These reactions build tissues.

For the design of his new device, Nocera relied on the help of scientists who specialize in photosynthesis and electrical engineers who work with silicon-based solar cells. “But most of all,” he says, “I relied on the discoveries of other chemists around the world … to design the roadmap to deliver the artificial leaf.”
About the size of a playing card, Nocera’s silicon solar cell is coated on each side with two different types of catalyst. Catalysts are materials that accelerate chemical reactions. In this case, they helped convert sunlight and water into fuel.
Here’s how it works: The cell is placed in a pail of water in full sunlight. There it splits the water into its two ingredients: hydrogen and oxygen. The hydrogen bubbles up
from one side of the cell; oxygen bubbles from the other. The gases are then captured and stored separately from each other and fed into a fuel cell to generate electricity when needed.
A fuel cell produces electricity by combining a fuel — usually hydrogen — with oxygen. Unlike batteries, fuel cells need a constant source of fuel and oxygen to run. As long as they are supplied with these two ingredients, fuel cells can keep producing electricity indefinitely.
Traditional solar cells generate electricity only when the sun is shining. Nocera`s advanced solar cell uses water and sunlight to make fuels — oxygen and hydrogen — that can be used anytime, even after the sun goes down. Whenever power is needed, that oxygen and hydrogen would be fed into a fuel cell. There the two elements would be combined back into water while delivering an electric current.
“With the artificial leaf — and as in photosynthesis — the sunlight is converted to a fuel, which can be stored,” explains Nocera. If fuels can be stored, they can be moved and used at will — not just when the sun shines. “So the artificial leaf is a step well beyond a solar cell,” he explains.
The artificial leaf is not a new concept. Other attempts at advanced solar cells used rare, expensive metals, he says, and had a life span of barely a day.
Nocera’s leaf is made of inexpensive and widely available materials, including catalysts made from nickel and cobalt. In a lab experiment, he showed that his leaf can operate nonstop for at least 45 hours without a drop in activity.
He now has plans to market the leaf. Nocera hopes that someday people living in areas with no electricity will be able to use his artificial leaf to do things like power lights in the evening so that students can do homework.
And maybe some of those students will one day make their own amazing biomimicry discoveries. In fact, Nocera has a bit of parting advice for anyone interested in doing what he, Gorb and Fish have done.
“You need to be inquisitive and totally dedicated,” he says. “Let your mind go. But you can only do so if you immerse yourself in a subject deeply. Then you can see what
others cannot.”
Power Words (adapted from the New Oxford American Dictionary)
molecule The combination of two or more atoms, rendering the smallest unit of any chemical. For instance, two hydrogen atoms make up a hydrogen molecule. Add an oxygen atom to that hydrogen molecule, and you now have a molecule of water.
entomology The study of insects.
hydrodynamics The study of liquids in motion.
photosynthesis The process by which green plants use sunlight to make food from carbon dioxide and water.
silicon A non-metallic element found in Earth’s crust. It can conduct electricity under some conditions. This makes it ideal for controlling electric currents, such as those in a solar cell.
catalyst A substance that triggers or speeds up a chemical reaction without itself being affected.
Word Find (click here to print puzzle)

This is one in a series on careers in science, technology, engineering and mathematics made possible by support from the Northrop Grumman Foundation.







